Carina Masters
In order to fully understand heat, a scientist must understand one of the most complex concepts in science. Atoms. Atoms are the little building blocks of life, impossibly small, and extremely difficult to study. They are invisible to the naked eye, and scientists still aren't 100% sure what an atom truly looks like. Scientists use different models of atoms to study different concepts.
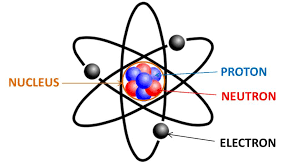
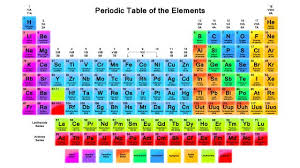
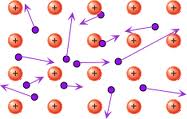
The basic understanding of atoms is that they are made up of even smaller particles called electrons, protons and neutrons. Electrons have a negative charge, protons have a positive charge, and neutrons have neutral, or no charge. These particles dictate many things about the atoms, and are what causes atoms to be one kind or another. They also make it so that an atom is an isotope, an atom with an unusual amount of neutrons, or an ion, an atom with an unusual number of electrons such as salt. Atoms are organized on a periodic table, showing their type, atomic number, electron configurations, and chemical properties. Electrons exist in the electron shell, or outer rings, separate from the nucleus of the atom, which holds the protons and neutrons. A molecule is created when two or more atoms bond together, usually through a shared electron, and a compound is a molecule made of different elements, such as water and carbon dioxide, but not oxygen.
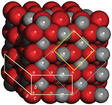
Atoms also determine if a molecule is liquid, gas, or solid. Atoms in a solid are tightly bonded, and don't move very much. Liquids are loosely bonded and move more quickly than a solid, and gases are not bonded and move extremely quickly.
The basic understanding of atoms is that they are made up of even smaller particles called electrons, protons and neutrons. Electrons have a negative charge, protons have a positive charge, and neutrons have neutral, or no charge. These particles dictate many things about the atoms, and are what causes atoms to be one kind or another. They also make it so that an atom is an isotope, an atom with an unusual amount of neutrons, or an ion, an atom with an unusual number of electrons such as salt. Atoms are organized on a periodic table, showing their type, atomic number, electron configurations, and chemical properties. Electrons exist in the electron shell, or outer rings, separate from the nucleus of the atom, which holds the protons and neutrons. A molecule is created when two or more atoms bond together, usually through a shared electron, and a compound is a molecule made of different elements, such as water and carbon dioxide, but not oxygen.
Atoms also determine if a molecule is liquid, gas, or solid. Atoms in a solid are tightly bonded, and don't move very much. Liquids are loosely bonded and move more quickly than a solid, and gases are not bonded and move extremely quickly.